
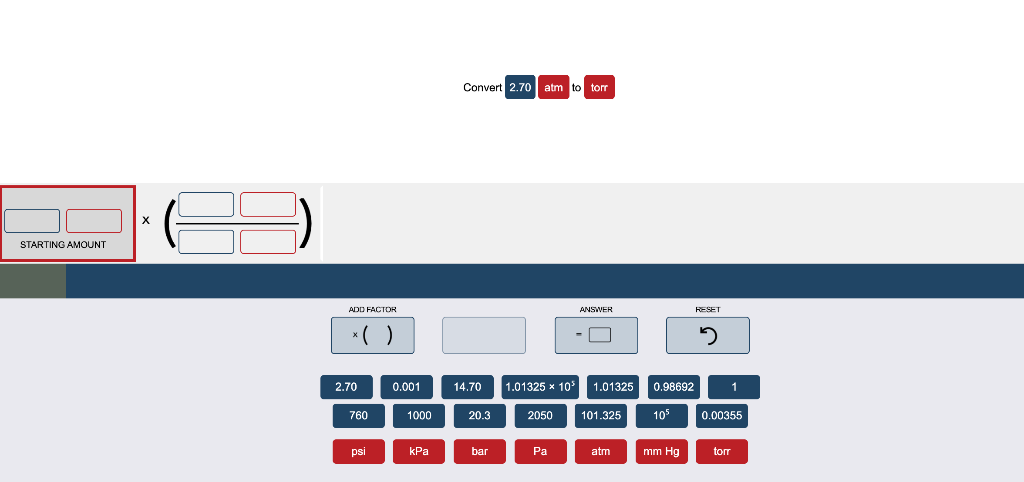
The mm Hg pressure gauge reads 1500 mm Hg this is approximately twice the 760 mm Hg needed to be 1 atm. So the 150 psi (~10 atm) is less than the 15 atm pressure gauge reading. In this question you have a 150 psi pressure on one of the gauges this is equal to about 10 times the 15 psi that is required to be equal to 1 atm. So when you use this thinking you realize that it will take 760 mm Hg to be equal to 1 atm. Similarly, a mm Hg unit is smaller still - about 1/760-th of an atm. multiply the atm value by 760 mmHg/atm or 0.92atm x 760.0 mmHg/atm the atm units cancel leaving mmHg Conversion of 735.5. In fact, a psi is roughly 1/15-th of an atmosphere. Since approximately 15 psi units are equivalent to 1 atm unit, a single psi unit must be rather small compared to a single atm unit.

This Game Plan begins by conceptualizing the size of an atm to that of a psi to that of a mm Hg. The second Game Plan (the one we are strong advocates of) is much more conceptual in nature. Once all three Pressure amounts are in atm units, you can easily compare to determine which is greatest, middlest, and smallest. Using this Game Plan, you would have to use the unit equivalencies (1 atm = 14.7 psi AND 1 atm = 760 mm Hg) to make two conversion factors and convert to Pressure in atm units. That is convert the pressure value in psi to atm and the pressure value in mm Hg to atm. Select a unit - like atmospheres - and convert the two non-atm amounts to atmospheres. The first Game Plan is to treat the question like a conversion problem. There are probably two basic game plans that a student could use to answer this question. in discussions of permeability to present a table of conversion factors. A good game plan equips you with a strategy for proceeding from the given information in the problem to the destination - the answer to the problem. Gas pressure is indicated in terms of mmHg, atmospheres, bars, or (for water. Like any problem in Chemistry, it is important to have a Game Plan.
in three different units and you have to compare them to determine the greatest, middlest, and smallest pressure value. For significant figure purposes, the standard pressure of 760 mmHg has three significant figures.In this question, you are given three pressures. The air pressure is about 80% that of standard atmospheric pressure at sea level. Use conversion factors from the equivalent pressure units to convert from mmHg to atm and from mmHg to kPa. Is 1 mmHg the same as 760 atm Pressure Units and Conversion. Step 1: List the known quantities and plan the problem. What is 1 atm equal to in mmHg 1 atm 101,325 Pascals 760 mm Hg 760 torr 14.7 psi. The atmospheric pressure in a mountainous location is measured to be 613 mmHg. Sample Problem: Pressure Unit Conversions To do so, we will use the equivalent standard pressures shown above. It is important to be able to convert between different units of pressure. The atmospheric pressure at sea level is 14.7 psi. Atmospheric pressure is also often stated as pounds/square inch (psi). The formula to convert from mmHg to atm is: atm mmHg 760 Conversion Example Next, let's look at an example showing the work and calculations that are involved in converting from millimeters of mercury to atmospheres (mmHg to atm). Standard atmospheric pressure is called 1 atm of pressure and is equal to 760 mmHg and 101.3 kPa. Another commonly used unit of pressure is the atmosphere (atm). A pascal is a very small amount of pressure, so the more useful unit for everyday gas pressures is the kilopascal (kPa). The pascal (Pa) is the standard unit of pressure. Convert two pressures from pound force per square inch to atmospheres.
An equivalent unit to the mmHg is called the torr, in honor of the inventor of the barometer, Evangelista Torricelli. This MATLAB function computes the conversion factor from specified input pressure. One unit of gas pressure is the millimeter of mercury (mmHg). Tire pressure is best measured when the tire is cold since driving the car for a while will heat up the air in the tire and increase the pressure.Ī barometer measures gas pressure by the height of the column of mercury. The pressure on the tire is the maximum pressure for that tire, not the recommended one.
Conversion factor for mmhg to atm manual#
The recommended pressure for that model of car (usually somewhere between 32-35 psi) is usually listed in the owner’s manual or stamped somewhere inside the door. The car gets better gas mileage and the tires don’t wear out as fast. The ride is smoother and safer than with lowered pressure. There are several benefits to maintaining the proper air pressure in a car tire.
